
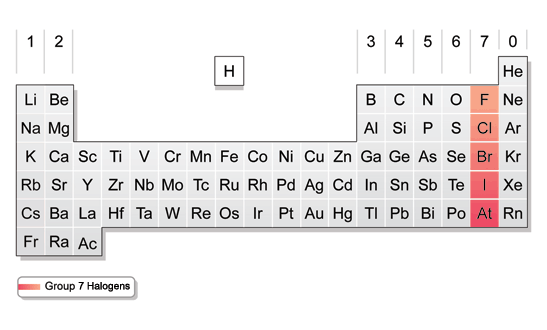
This is the case for all of the halogens. As in fluorine, each of the electrons in chlorine’s outer shell only feels a charge of +7. Ten of these electrons are in inner shells, shielding the charge of ten protons. Chlorine has seventeen protons and seventeen electrons. They shield the charge of two of fluorine’s protons, so each electron in fluorine’s outer shell only feels a charge of +7. Fluorine has nine protons and nine electrons - two of these electrons are in an inner electron shell. Remember that electronegativity is an atom’s ability to attract a shared pair of electrons. Knowing what you know about atomic radius, can you predict the trend in Electronegativity as you go down the halogen group? Take a look at Polarity if you need a reminder.Īs you move down the group in the periodic table, the halogens decrease in electronegativity. Let's look at some trends in chemical properties within the halogen group. Because both alkanes and halogen molecules are nonpolar, the attractions broken between two halogen molecules are roughly equal to the attractions formed between a halogen molecule and an alkane molecule - so they mix readily. Solubility is all to do with the energy released when molecules in a solute are attracted to molecules in a solvent. Halogens are much more soluble in inorganic solvents like alkanes. There is no point even considering the solubility of fluorine - it reacts violently the instant it touches water!
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
